 
To explain his hypothesis, he took the example of sodium and chlorine atoms. Lewis hypothesized that atoms achieve a stable octet because of the linkage of chemical bonds. This octet of electrons acts for a specifically stable electronic order. In the occurrence of a noble gas, all the cube’s eight corners are occupied.ĭue to the arrangement of eight electrons, it is termed the octet of electrons. This way, a single valence shell electron of an atom (suppose sodium) would occupy one corner of the cube. He supposed that these eight electrons inhabited the corners of a cube surrounding the ‘Kernel’. He assumed that the outer shell of the ‘Kernel’ accommodates a maximum of eight electrons. 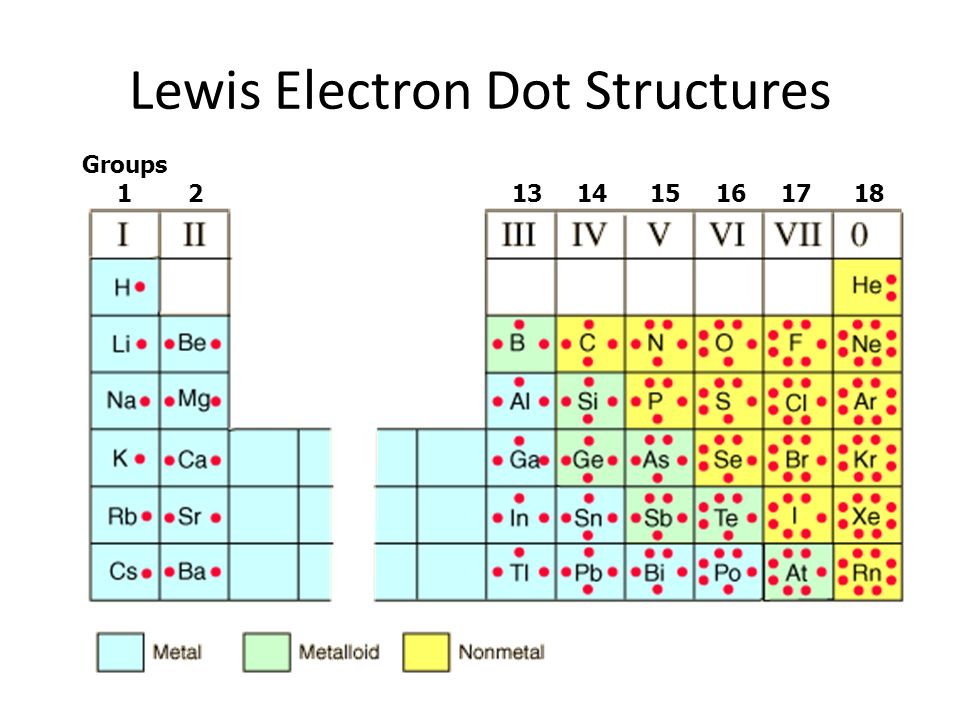
This ‘Kernel’ includes an atom’s nucleus and inner electrons. Lewis, an American chemist, took the atom as a positively charged ‘Kernel’. They provided some logical descriptions by experimenting with the inertness of noble gases. In 1916 a satisfactory explanation was declared when Lewis and Kössel succeeded in this experiment independently. Several attempts are made to explain the chemical bond formation in terms of electrons. The following article describes more about the ‘Lewis Dot Structures.’ Lewis introduced an article, ‘The Atom and the Molecule.’ It explained the idea of forming chemical bonds by sharing electrons with the concept of electron dot structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2023
Categories |

 RSS Feed
RSS Feed
